In the realm of emergency medical solutions, innovation has often been the key to saving lives. In collaboration with the esteemed regulatory authorities in Thailand and our dedicated team of experts, we are proud to present the Sonmol Rescue Device / Heimlich Device. This groundbreaking device promises to redefine the landscape of choking rescue procedures.
Heimlich Device Successfully Registered in Thailand
We are thrilled to announce a monumental achievement for Sonmol’s innovative line of emergency medical devices. Our Heimlich Device, known as the Choking Rescue Device, has received successful registration in Thailand, thanks to the collaborative efforts of our partner and the regulatory authorities.
The significance of introducing our Choking Rescue Device to more countries cannot be overstated. Choking incidents can occur anytime, anywhere, and having access to effective rescue devices is crucial for saving lives. By expanding the reach of our device to new regions, we are empowering communities with the tools they need to respond swiftly and effectively to choking emergencies.
In many parts of the world, access to advanced medical equipment may be limited, especially in rural or underserved areas. Introducing the Sonmol Choking Rescue Device to more countries helps bridge this gap, ensuring that life-saving technology is available where it’s needed most. Whether in homes, schools, restaurants, or healthcare facilities, every individual deserves access to reliable emergency medical devices.
Moreover, raising awareness about the importance of choking rescue procedures is essential for promoting public safety and reducing preventable fatalities. By introducing our device to more countries, we are not only providing a tangible solution but also advocating for proactive measures to prevent choking incidents and educate communities about proper emergency response protocols.
The successful registration of the Sonmol Choking Rescue Device in Thailand marks a significant step forward in our mission to enhance global healthcare standards. We are committed to continuing our efforts to introduce this life-saving technology to even more countries, ensuring that individuals worldwide have the best possible chance of surviving choking emergencies.
You can find our Sonmol Choking Rescue Device listed on the Thailand Food and Drug Administration’s website. Simply visit this link and search with the keywords “Sonmol Choking Rescue Device” to access detailed information about our product’s registration status.
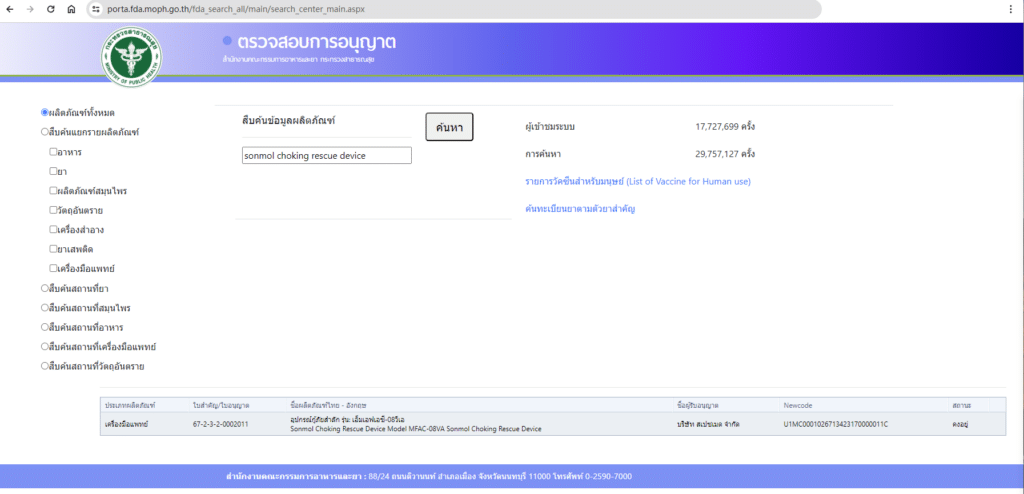
Our dedication to enhancing global healthcare standards is evident in the development and registration of the Sonmol Choking Rescue Device. This achievement not only highlights our commitment to innovation but also emphasizes our mission to ensure swift and effective response to choking incidents worldwide.
If you are interested with our Thailand distributor or partner, visit their website by clicking HERE.
Call to Action:
As we celebrate this significant milestone in Thailand, we extend an invitation to distributors and partners in other countries to join us in our mission to make life-saving technologies accessible worldwide. Priority for first aid device distributors, specifically, cannot be emphasized enough.
If you are a distributor or know of one who shares our commitment to enhancing global healthcare standards, we encourage you to reach out to us. By collaborating with Sonmol, you can play a pivotal role in bringing our Choking Rescue Device to communities around the world, saving lives and making a lasting impact on public health.
Together, let’s make choking rescue procedures more accessible, ensuring that no life is lost to a preventable choking incident. Contact us today to explore partnership opportunities and become part of our global network dedicated to advancing emergency medical solutions.
Contact Us via WhatsApp




